 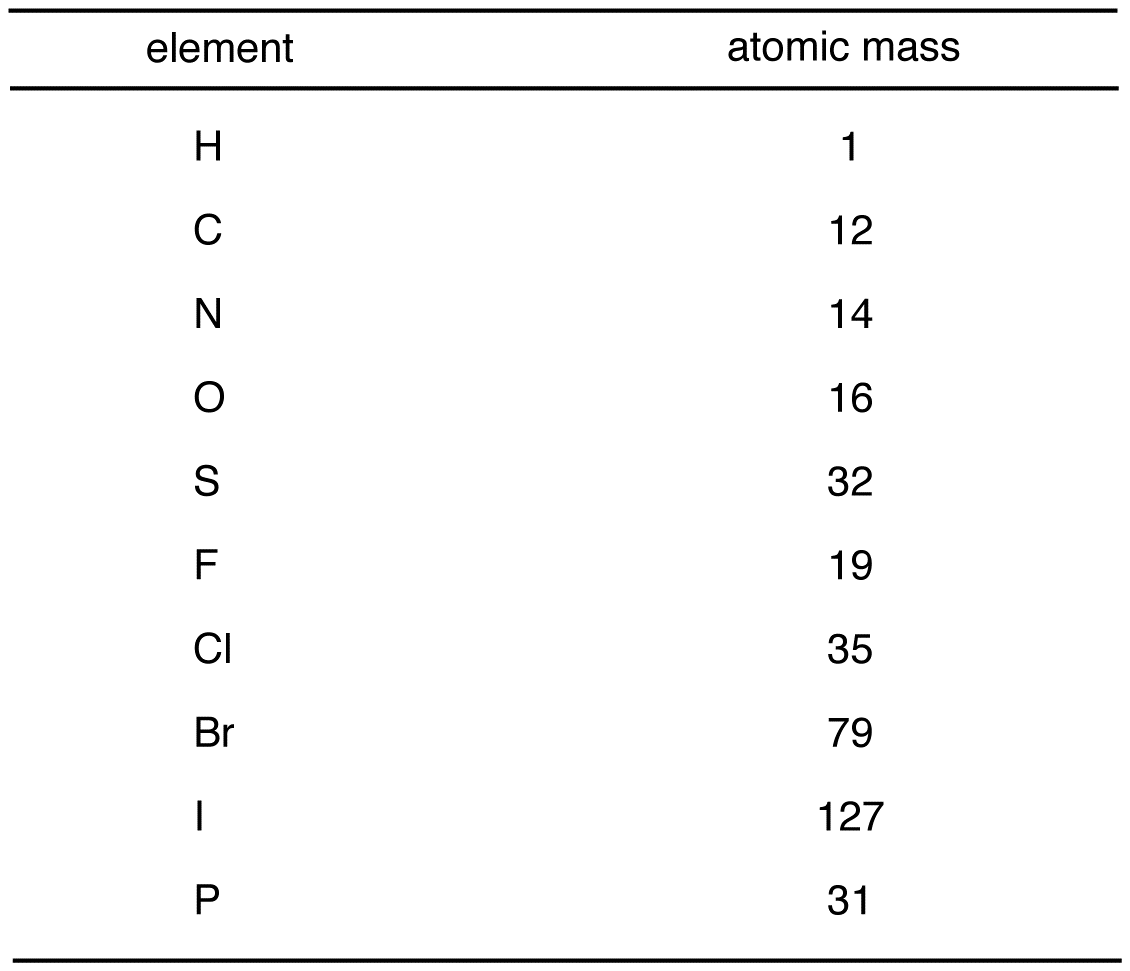
For comparison, the atomic mass of a carbon-12 atom is exactly 12 daltons. Equivalently, the relative isotopic mass of an isotope or nuclide is the mass of the isotope relative to 1/12 of the mass of a carbon-12 atom.įor example, the relative isotopic mass of a carbon-12 atom is exactly 12. The relative isotopic mass, then, is the mass of a given isotope (specifically, any single nuclide), when this value is scaled by the mass of carbon-12, where the latter has to be determined experimentally. This loss of units results from the use of a scaling ratio with respect to a carbon-12 standard, and the word "relative" in the term "relative isotopic mass" refers to this scaling relative to carbon-12. While atomic mass is an absolute mass, relative isotopic mass is a dimensionless number with no units. Relative isotopic mass (a property of a single atom) is not to be confused with the averaged quantity atomic weight (see above), that is an average of values for many atoms in a given sample of a chemical element. The atomic mass of atoms, ions, or atomic nuclei is slightly less than the sum of the masses of their constituent protons, neutrons, and electrons, due to binding energy mass loss (per E = mc 2). The dimensionless (standard) atomic weight is the weighted mean relative isotopic mass of a (typical naturally occurring) mixture of isotopes. Because substances are usually not isotopically pure, it is convenient to use the elemental atomic mass which is the average ( mean) atomic mass of an element, weighted by the abundance of the isotopes. The atomic mass of an isotope and the relative isotopic mass refers to a certain specific isotope of an element. The sum of relative isotopic masses of all atoms in a molecule is the relative molecular mass. Thus, the atomic mass of a carbon-12 atom is 12 Da by definition, but the relative isotopic mass of a carbon-12 atom is simply 12. The relative isotopic mass (see section below) can be obtained by dividing the atomic mass m a of an isotope by the atomic mass constant m u yielding a dimensionless value. Conversion between mass in kilograms and mass in daltons can be done using the atomic mass constant m u = m ( 12 C ) 12 = 1 D a is the experimentally determined molar mass of carbon-12. Thus, the numeric value of the atomic mass when expressed in daltons has nearly the same value as the mass number. The protons and neutrons of the nucleus account for nearly all of the total mass of atoms, with the electrons and nuclear binding energy making minor contributions. 1 Da is defined as 1⁄ 12 of the mass of a free carbon-12 atom at rest in its ground state. Although the SI unit of mass is the kilogram (symbol: kg), atomic mass is often expressed in the non-SI unit dalton (symbol: Da) – equivalently, unified atomic mass unit (u). Multiply the number of each atom by its atomic weight (found on the Periodic Table of the Elements).The atomic mass ( m a or m) is the mass of an atom. Water is made up of 2 hydrogens and 1 oxygen.Ģ. The molar mass is calculated by taking the sum of the atomic weights of all the atoms which form the molecule.įirst determine the number of each atom in the formula.ġ. When there is parenthesis's around a group of atoms we must multiply each atom by the subscript to get the total number of each element.Ĭa(NO 3) 2 1 Calcium 2 Nitrogen and 6 Oxygen -> 3 x 2 Reading Compounds that have (parenthesis's). If there is only 1 atom we do not use subscripts. The subscript belongs to the element it follows and indicates how many of that atom there are. Reading Simple Chemical Formulas- Each element begins with a CAPITAL letter. The units are atomic mass units ( amu ).ġamu =1.660 538 782×10 –27 kg 1 amu is 1/12 the mass of a carbon-12 atom which has a mass of 12.0000. Molecular Mass is the mass of a given molecule (NOT MOLES of molecules). 
Molecular Weight is the molar mass of a COVALENT compound. The unit for molar mass (note it is the mass of a mole) is grams/mole.Ītomic Weight is the molar mass of an element.įormula Weight is the molar mass of an IONIC compound. Molar Mass is the mass of one mole of a substance (6.02 x 10 23 formula units). (Molar Mass, Molecular Weight, Gram Formula Mass)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
